


 Français
Français


The terms “antibiotics” and “antimicrobials” are sometimes used interchangeably. ”Antimicrobial” is a broad term to describe medicines that can kill or block the growth of various microorganisms including bacteria, viruses, fungi, and parasites. Antibiotics are a specific type of antimicrobial = used to treat infections caused by bacteria. Worldwide, there is concern about antimicrobial resistance (AMR) and the prudent use of all antimicrobial drugs.
Residues are not the Issue
While industry needs to remain vigilant about preventing antibiotic residues, antibiotic residue is not the present-day concern associated with antimicrobial use.
Antibiotic residues refer to the presence of the antibiotics themselves in the final meat product. The Canadian Food Inspection Agency monitors for residue levels and chicken has maintained an excellent track record. This is because of chicken farmers’ excellent track record in adherence to withdrawal times.
The Issue is Resistance
Antimicrobial resistance means that the antimicrobial drugs that used to be effective against a particular microbe no longer work because the microbe’s biological makeup has changed; it has become resistant to the treatment. The problem can occur naturally, or when an infection is treated with an antimicrobial which kills only some of the microbes1. Some bacteria are naturally resistant, such as certain types of bacteria being unaffected by penicillin. Other bacteria develop resistance by genetic mutations that allow them to survive in the face of environmental pressures, such as the presence of antibiotics, or by receiving resistance genes transferred from other bacteria.
Cross-resistance can also develop because of the similarities among different antimicrobials. For example, tylosin and erythromycin are two different antibiotics used in animals (erythromycin is also used in people). Development of resistance to tylosin in previously susceptible bacteria can also confer resistance to erythromycin in the same bacteria.
Antimicrobial use is a major driver for the development of antimicrobial resistance. Bacteria adapt and become resistant when they are exposed to antibiotics used by humans and in agriculture – thus the pressure to reduce antibiotic use.
Antibiotic resistance – its development and how it is shared amongst bacteria – is a complex process, but one that can have significant negative impacts on human and animal health.
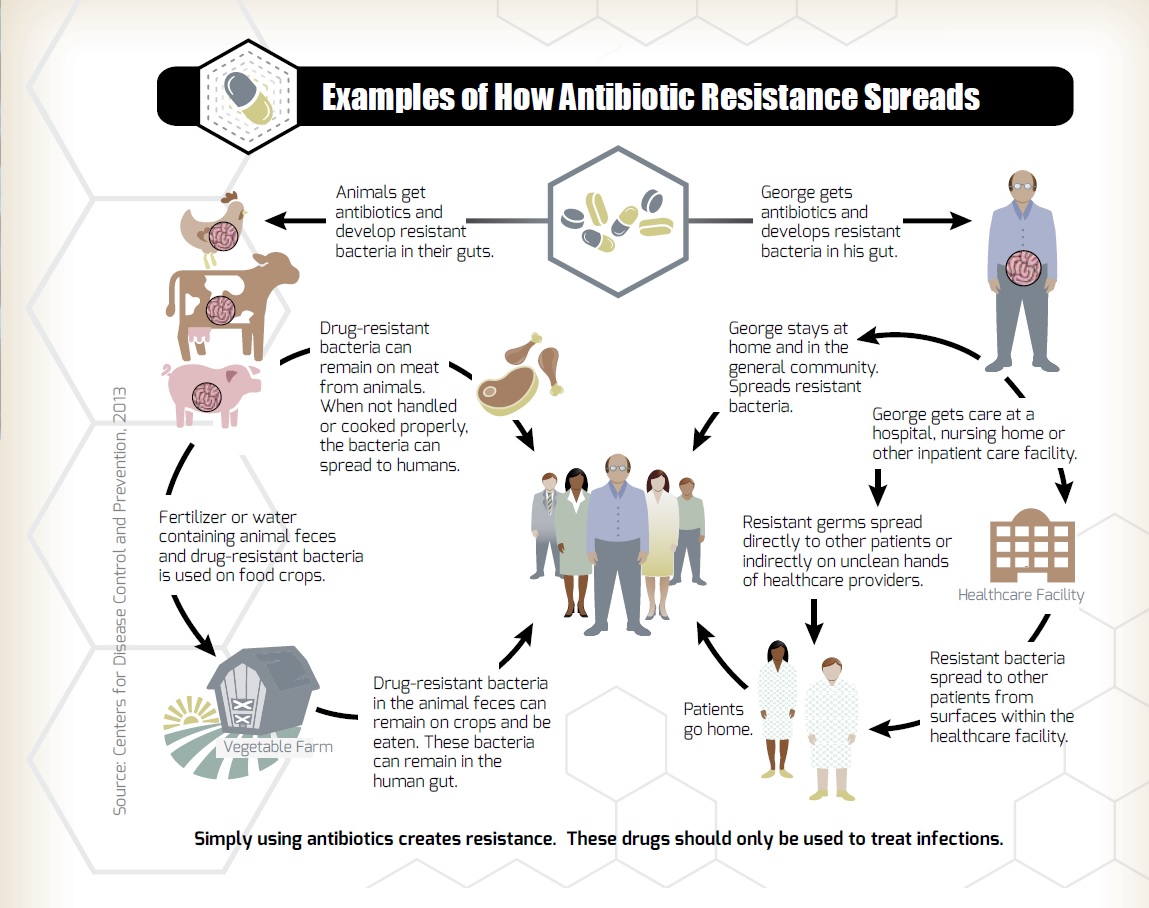
The diagram from the (U.S.) Centers for Disease Control and Prevention (CDC), outlines how resistance can develop and spread among the community.
Resistance is a Threat to Public Health and to Agriculture
The World Health Organization (WHO) has stated that antimicrobial resistance is a serious threat to global public health that threatens the effective prevention and treatment of infections caused by bacteria and other microorganisms. Without effective antimicrobials, infections will become untreatable and illnesses will become longer and more severe.
This threat is amplified by the fact that very few new antibiotics have been discovered in recent decades – which enforces the need to preserve the effectiveness of current antibiotics.
The threat to public health has been well documented, both in Canada and internationally:
The threat to agriculture is similar – increased resistance will prevent the use of antibiotics for treatment of disease, while threatening both the welfare of animals and the food supply.
Does Antibiotic Use at the Farm Contribute to This Problem?
All antibiotic use can impact and help develop resistance, whether in humans or in agriculture.
Evidence that antibiotic use in food animals can result in antibiotic-resistant infections in humans has existed for some time and has been detected in observational studies as well as in randomized trials5. When antibiotics are used in agriculture, there is an opportunity for bacteria to develop resistance. As shown in the figure, improper handling and cooking of poultry products can result in people becoming ill from bacteria that may be present on poultry meat (e.g. Salmonella). If those bacteria are antibiotic-resistant, then treatment of the resulting illness can become much more difficult. There is also a possibility of drug-resistant bacteria in animal feces coming in contact with the environment and allowing the potential spread of resistance genes that way.
Research appears to show that the risk for farm workers and families may be more elevated than the general public, due to their closer association with live animals. Research is indicating that farm workers and families have higher antimicrobial resistance patterns than the general population6.
While any antibiotic use can lead to resistance, it is also encouraging to know that prudent use policies can have positive impacts on resistance levels. For example, surveillance data from the Canadian Integrated Program for Antimicrobial Resistance Surveillance (CIPARS) has shown reduced resistance levels across antimicrobial classes as a result of Chicken Farmers of Canada’s AMU reduction steps7.
The International Effort to Maintain the Effectiveness of Antibiotics
In 2015, the World Health Organization issued a Global Action Plan on AMR developed with both the World Organization for Animal Health (OIE) and the Food and Agriculture Organization, which has resulted in a number of initiatives to reduce use around the world8.
For example, in April 2016, G7 Agricultural Ministers made a declaration that all members were encouraged to ensure the prudent use of antibiotics, to phase out antibiotics for growth promotion and to preserve the use of antibiotics only for therapeutic reasons. This was followed in September 2016 by the United Nations General Assembly adopting a political declaration aimed at combating the global threat posed by antimicrobial resistance.
In the US, as a result of reduced AMU declarations by several different poultry companies, there has been growth in NAE production (no antibiotics ever) over the last several years. The percentage of chickens raised in NAE systems was the highest in 2019 at 58%, and dipped slightly in 2020 to 52%9. Some of the producers moving away from NAE switched to an “ionophore-only” type of program, indicating that the commitment to reducing antibiotics is still strong9.
In Canada in 2017, the Canadian government released the Pan-Canadian Framework on AMR and AMU, calling for action by all sectors on the issue. The framework focusses on surveillance, infection prevention and control, stewardship, and research and innovation and CFC’s Responsible AMU Strategy aligns with these key areas.
Most recently, the European Union (EU) is set to ban certain antimicrobials for any use in food animal production as early as January 2022. The focus is on antimicrobials important in human medicine, and this could have far-reaching implications for trade and precedence-setting for AMU worldwide.
References: